Both substances increase the risk of engaging in unsafe sexual practices, which can lead to the transmission of sexually transmitted infections (STIs) and other health complications. Users report feeling more affectionate and emotionally open, which can lead to unprotected and potentially risky sexual behaviors due to lowered inhibitions. MDMA, often preferred for its empathy-enhancing and euphoric effects, is commonly used in chemsex contexts to deepen feelings of emotional intimacy and connectivity with sexual partners. Over time, the brain comes to rely on the drug to stimulate feelings of happiness and pleasure, resulting in physical dependence. Symptoms of addiction to MDMA and MDA include continued use despite adverse side effects, tolerance (where larger doses are needed to achieve the same effect), withdrawal symptoms, and cravings.
DMA derivatives are a prevalent building block of natural products and bioactive molecules used in materials, medicines, and agrochemical industries. To avoid the formation of NDMA from DMA, it’s essential to control factors that promote its synthesis, such as nitrites, reaction conditions, and storage conditions. It is not intentionally added to products but can emerge as a by-product of specific chemical reactions. DMA has a hydrophilic (water-attracting) nature, due to the presence of the nitrogen atom, and the ability to form hydrogen bonds with water molecules.
Healthcare Professionals
The Texas Group advertised “Ecstasy parties” at bars and discos, describing MDMA as a “fun drug” and “good to dance to”. Starting in 1983, the Texas Group mass-produced MDMA in a Texas lab or imported it from California and marketed tablets using pyramid sales structures and toll-free numbers. Hoping MDMA could avoid criminalization like LSD and mescaline, psychotherapists and experimenters attempted to limit the spread of MDMA and information about it while conducting informal research. Psychotherapists who used MDMA believed the drug eliminated the typical fear response and increased communication.
Moreover, drug effects depend on factors like dosage, purity, and setting. As a result, people may experience negative psychological aftereffects such as depression, anxiety, and fatigue, which can persist for several days following MDMA use. Among these, MDMA exerts its most potent effects on serotonin. Similar to MDMA, MDA’s impact on serotonin receptors can lead to depression, anxiety, and fatigue.
10 Venlafaxine Hydrochloride
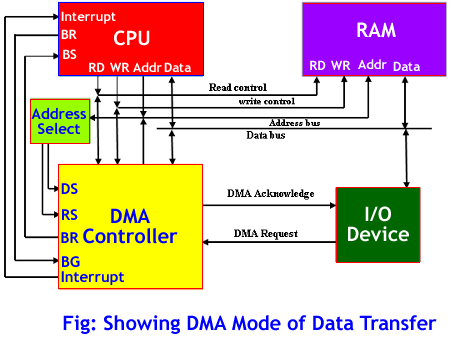
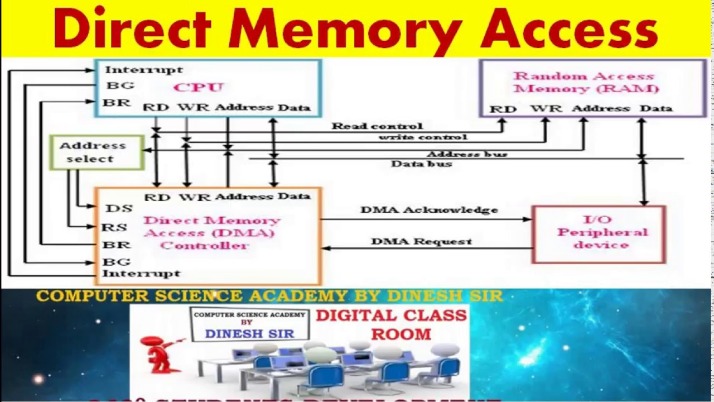
DMA inhibits the binding activity of BRD2 and BRD4 (Fig. 1a,c) with an IC50 value of 11 mM and 6 mM, respectively (Fig. 1b,d). Busulfan is a DNA-alkylating drug applied as a myeloablative chemotherapeutic agent prior to stem cell transplantation in pediatric and adult patients suffering from neuroblastoma or other malignancies15. In high risk neuroblastoma treatment DMA, at high concentration, is administered to facilitate busulfan application14. Organic solvents are commonly used in the pharmaceutical industry as reaction media, for product synthesis and as drug-vehicle.
What Are The Effects Of DMT?
Diphenhydramine citrate is an antihistaminic drug, used to treat allergies, hay fever, insomnia, and common cold-like symptoms.76 It was discovered in 1943 by George Rieveschl and it became the first prescription antihistaminic drug approved by FDA in 1946. After that, the cyano base of compound 117 is subsequently decyanated with flakes of caustic potash (KOH) to produce chlorphenamine 118 with a good yield. It is synthesized by reacting 2-chloropyridine 114 with 2-(4-chlorophenyl) acetonitrile 115 in the presence of NaNH2 in O-xylene to give compound 116, which undergoes condensation with 2-chloro-N,N dimethylethanamine at room temperature for 8 h to give compound 117. Chlorpheniramine maleate (CPM) is a first-generation antihistaminic agent, classified as an alkylamine derivative, and acts by antagonizing the H1 receptor.71 It is employed in the treatment of allergies, hay fever, and common cold. On reacting compound 112 with dimethylamine, the target molecule trimeprazine 113 was formed70 (Fig. 28). The synthesis of trimeprazine is started with the reaction between phenothiazine 110 and methacrylonitrile to produce compound 111, which on reduction with LiAlH4 followed by alkylation with methanesulfonyl chloride (MsCl) to from compound 112.
Other Potential Treatment Applications
It is present in cohoba, a hallucinogenic drug derived from the seeds of Piptadenia peregrina. More detail and supporting information is in the main article. Despite its illegal status, people sometimes use DMT in religious ceremonies and various settings for an “awakening” or to obtain deep spiritual insight. The substance has a high potential for abuse, no government-recognized medical use, and a lack of accepted safety parameters for use.
According to one participant in an ethnographic study, the Texas Group produced more MDMA in eighteen months than all other distribution networks combined across their entire histories. In response to the proposed scheduling, the Texas Group increased production from 1985 estimates of 30,000 tablets a month to as many as 8,000 per day, potentially making two million ecstasy tablets in the months before MDMA was made illegal. Sensational media attention was given to the proposed criminalization and the reaction of MDMA proponents, effectively advertising the drug. Bill Mandel reported on “Adam” in a 10 June San Francisco Chronicle article, but misidentified the drug as methyloxymethylenedioxyamphetamine (MMDA).
This is analogous to other findings in which various 2C and NBOMe drugs have been found to produce dopaminergic elevations and reinforcing effects in rodents. 2,5-DMA shows dramatically reduced potency as a serotonin 5-HT2A receptor agonist compared to the DOx drugs, such as 2,5-dimethoxy-4-methylamphetamine (DOM). Some people also just tend to metabolize certain drugs more rapidly or more slowly than others.
Drug Used In Treatment Of Peptic Ulcer
These risks are exacerbated by physical activity, such as dancing for extended periods in hot, crowded environments, which is common at raves and music festivals where these drugs are frequently used. The stimulant effects of these drugs can lead to cardiac complications, such as arrhythmias, myocardial infarction, and in extreme cases, heart failure. MDA, with its higher affinity for serotonin receptors, is considered particularly potent in this regard, potentially leading to more severe neurological consequences than MDMA. Known formally as 3,4-methylenedioxymethamphetamine (MDMA), and 3,4-methylenedioxyamphetamine (MDA), they can lead to a wide range of side effects and pose severe health risks if abused.
Article Details
- Therapeutic agents targeting protein expression on the epigenetic level have shown encouraging antitumor activity.
- Some athletic associations, courts, and drug abuse treatment centers also require drug testing.
- MDMA tablets typically have low purity due to bulking agents that are added to dilute the drug and increase profits (notably lactose) and binding agents.
- 3,4-DMA produces 3-methoxy-4-hydroxyamphetamine (MHA) as its major metabolite in dogs and monkeys.
- Compound 268 was then combined with sodium methoxide in methanol and treated with trimethyl phosphate at 70 °C to yield compound 269.
We found ovariectomy promoted a significant increase in body mass (Fig. 2a) and enhanced marrow lipid accumulation within bone marrow (Fig. 2b) while DMA reversed these effects. In bone, inhibition of BRD4 stops differentiation to osteoclasts21,22, the cells responsible for bone degradation. A substantial portion of the bioactivity of the excipient NMP in terms of bone resorption and inflammation can be linked to its affinity to bromodomains18,19. This study focuses on the potential therapeutic benefits of DMA in osteoporosis, bone regeneration and related bone diseases, and emphasizes the fact that studies where DMA or other solvents were used as controls must be analyzed with caution. DMA is FDA approved as excipient and therefore widely used as drug-delivery vehicle13.
- It is unclear why Shulgin provided information about DOM to Stanley, since doing so had the potential to risk Shulgin’s professional career and the DOET clinical studies.
- Due to its selectivity, DOB is often used in scientific research when studying the 5-HT2 receptor subfamily.
- WebMD does not provide medical advice, diagnosis or treatment.
- The cyclobutane ring of compound 162 was then opened to form an o-quinone methide intermediate, which underwent a Diels–Alder cycloaddition with compound 161, resulting in the formation of the endo adduct 163.
- DMA is a weak base and it reacts with acid to form salts such as dimethylamine hydrochloride.2 DMA is present in a broad variety of animals and plants and can be found in many foods at low concentrations.3 DMA is a precursor of many industrially important compounds.
Fig 19 Synthesis Of Thiothixene
The loan pair of nitrogen atoms of DMA participates in many nucleophilic reactions, for example bis(dimethylamino)methane was formed when DMA reacted with formaldehyde. DMA is the source of the solvents such as dimethylformamide and dimethylacetamide. It is a colourless gas that, at lower concentrations, smells like fish and, in higher concentrations, smells like ammonia.1 DMA gas is corrosive and easily dissolves in water to produce combustible solutions that are corrosive. Further research and development efforts are warranted to explore their full therapeutic capabilities and optimize their clinical utility in the treatment of various diseases. Therapeutically, DMA derivatives have shown promise in the treatment of infectious diseases, especially bacterial infections. Synthetic strategies for the preparation of DMA derivatives vary depending on the desired biological activity and target molecule.
Fig 40 Structure Of Chlortetracycline Hydrochloride
The condensation of compound 300 with 4-(4-chlorophenyl)-4-piperidinol 303 form loperamide hydrochloride 304. It is synthesized by reacting cyclohexanone 290, paraformaldehyde 291 and dimethylamine hydrochloride 292 (Mannich reaction) to give 2-dimethylaminomethyl-cyclohexanone hydrochloride 293, which reacts with NaOH to give intermediate 294. It works by two mechanisms, first is inhibition of the reuptake of serotonin and norepinephrine and second, it acts as an agonist of the μ-opioid receptor.
Fig 15 Synthesis Of Levomepromazine
BMP2-induced ALP activity was decreased by TNF-α treatment. TRAP activity (c) was determined after 6 days of treatment. Number of multinucleated cells (MNCs) (b) were determined after 6 days of treatment. Therefore, DMA enhanced bone regeneration and bone formation in vivo. Significantly more bone formed in this non-critical size defect when the membrane was loaded with DMA, compared to membrane alone (Fig. 4d,e).
They can also inject the drug, but this method has additional risks. Sometimes the drug can have a yellowish or pinkish color. Synthetic DMT usually comes in the form of a white, crystalline powder. Observational studies suggest it could have potential usage in depression, stress, and anxiety The latter contains DMT while the former contains MAOIs, which prevent certain enzymes in your body from breaking down DMT.